Metolazone Tablets
- Metolazone 2.5MG Tablet 100 Count (76385-0136-01)
- Metolazone 5MG Tablet 100 Count (76385-0137-01)
- Metolazone 10MG Tablet 100 Count (76385-0138-01)



Etodolac IR Tablets
- Etodolac IR 400MG Tablet 100 Count (76385-0118-01)
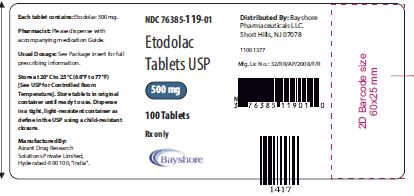
- Etodolac IR 500MG Tablet 100 Count (76385-0119-01)

Ketotifen Fumarate Ophthalmic Drops
- KETOTIFEN FUMARATE 0.035% 5ml (76385-0106-17)


